There seems to have been a whirlwind of activity around copper on some other forums,
http://www.reeffrontiers.com/forums/showthread.php?t=15666
http://www.reefland.com/forum/marin...tment/18835-copper-absorbs-into-glass-no.html
and I was asked to comment on a couple of threads at different forums, so I thought I'd also post that info here:
Here are my thoughts on copper:
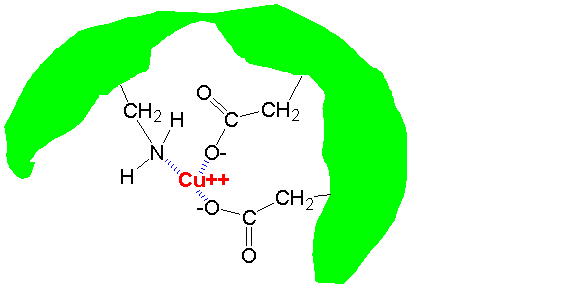
1. Copper in a reef aquarium is going to be strongly bound to organic materials. That is known to be true in natural seawater at normal copper levels. Maybe when you initially blast seawater with unbound copper there is substantial free copper for some period of time, until the copper concentration eventually drops, or the free copper has a chance to find organics to bind to.
2. While free copper may or may not bind much to glass (and likely none to silicone, IMO), organic bound copper will bind to both glass and silicone surfaces. Not into it, but onto it. It would be an interesting experiment to put some clear cured silicone into a copper/water solution to see if it turns blue. My expectation is that it will not, but I've not done the experiment.
3. It is my opinion that it is not too difficult to remove copper from all plastic and glass objects in an aquarium, but specifically not those that are calcium carbonate. A little bleach will remove organics and copper along with them. A little acid (like vinegar) will remove inorganic copper and some of its precipitates (like copper carbonate or oxide/hydroxide). I think all such objects can be reused after such cleaning.
4. Calcium carbonate surfaces are a much bigger concern. Just as magnesium gets onto and actually into calcium carbonate surfaces, copper and other similar ions would be expected to do the same thing. It is not trivial to clean live rock and sand. In most cases, I'd recommend tossing it. If that is just not an option, cleaning it in acid so that you actually dissolve some of the surface away is a reasonable thing to try, and some folks in my forum have done that.
"Why all the countless years have we not seen dying inverts in tanks where we used copper medications?"
I discuss copper toxicity here:
Tap Water in Reef Aquaria
http://www.advancedaquarist.com/issues/jan2004/chem.htm
from it:
"Toxic Metals in Tap Water: Copper
One of the biggest concerns for aquarists considering the use of tap water is copper. Copper is more toxic to marine invertebrates than it is to humans, so the EPA allows more in tap water than an aquarist would want in an aquarium. Specifically, the EPA has an action level at 1.3 ppm copper in tap water, and it is only a big problem (to the EPA) if more than 10% of the homes on the water supply exceed that level. For that reason, water supplies typically report the 90% level, meaning that 10% of homes exceed that stated value. Many also report the maximum level in any home tested.
Table 5 shows copper levels for a small selection of homes for all of the cities in this study. The reason that copper must be tested at the home tap is that most of the copper in public water supplies comes from the pipes in the home itself. Further, newer does not necessarily mean better, as fresh copper pipes might be even more inclined to release copper to the water than those that have had decades to build up protective coatings.
How much copper is too much? That is difficult to say, and certainly varies from organism to organism. In a recent test,5 Ron Shimek added copper to natural seawater and looked at the effect on sea urchin larvae. He found that concentrations above 10 ppb decreased the larval survival after 48 h, and that concentrations above 100 ppb killed all of them. Whether that translates into particular copper levels to be concerned about in typical reef aquaria is open to discussion. Nevertheless, it points out that copper is potentially toxic at levels well below the EPA action level of 1.3 ppm. That hypothesis is well supported by the literature on copper toxicity.6 Canadian Water Quality Guidelines for the Protection of Aquatic Life suggest that copper be kept to less than a few ppb in fresh water, but they do not provide a salt water recommendation. "
In terms of chemistry of inorganic copper absorption, bare glass is likely to carry a net negative charge in seawater (from silicate) and can bind copper as a copper silicate complex. This is well established in the scientific literature:
Adsorption of some divalent cations from aqueous solution on precipitated silica. Bye, Gerald C.; McEvoy, Michael; Malati, Mounir A. Mid-Kent Coll. Higher Further Educ., Chatham, UK. Journal of the Chemical Society, Faraday Transactions 1: Physical Chemistry in Condensed Phases (1983), 79(10), 2311-18.
Abstract
The adsorption of Co2+, Ni2+, Cu2+, Zn2+, and Cd2+ on the K+ form of a pptd. silica is reported. The adsorption isotherms of Co2+ and of Ni2+ at pH 7.0 and different temps. follow the Langmuir isotherm. The endothermic apparent heats of adsorption, 2 and 10 kJ/mol, resp., are compared with those of Cu2+ and Cd2+. The limiting amt. adsorbed, Xm, of the 4 ions decrease as the pH is lowered. An ion-exchange mechanism between hydrated K+ ions in the outer Helmholtz layer and metal, M2+, ions in soln. is proposed. The results also suggest that (MOH)+ ions are adsorbed at the higher pH values. A plot of Xm at pH 6.0 and 323 K for the 5 ions studied against at. no. exhibits the Irving-Williams sequence. At pH 3.0, i.e. close to the point of zero charge of SiO2, the adsorption results were used to calc. the free energy of specific adsorption, DG, using the Grahame equation. The DG values were -11.1, -10.7, -13.4, -13.3, and -12.1 kJ/mol for Co, Ni, Cu, Zn, and Cd, resp. Specific adsorption is tentatively ascribed to the formation of surface coordination complexes.
Ion-exchange adsorption of zinc and copper ions on silica. Kozawa, A. Univ. Nagoya, Japan. Journal of Inorganic and Nuclear Chemistry (1961), 21(3/4), 315-24.
Abstract
Ion-exchange adsorption of Zn and Cu ions on silica were examd. in 2M NH4Cl contg. ZnO or ZnCl2 and in M NH4OAc contg. 0.05M CuCl2, resp. The ion-exchange adsorption attains equil. in about 2 hrs. at 25°. The adsorption of Zn++ increases with the pH of the soln. up to pH 7.12, then decreases. Between pH 5.48 and 6.38 the mole-ratio of H+ released Zn2+ adsorbed is const. at .apprx.3.0. Zn++ adsorption on heated silica decreases markedly with increase in the heating temp. For Cu++ adsorption, the mole-ratio, H+ rel./Cu2+ ads., is 4.0. The infrared absorption spectra of silica samples, heated silica, and silica treated with Zn2+, Cu2+, and Na+ solns., were taken. A decrease in the absorption intensities for the bending vibration of OH groups on the silica is discussed. A surface complex mechanism for metal ion adsorption of silica is proposed; the silica surface is acting as tridentate ligands for the Zn++ adsorption. A new method for detg. the surface acid and the surface area of silica is suggested.
Reaction of silica gel with copper ions in acetate solutions. II. Rak, R. V.; Dushina, A. P.; Aleskovskii, V. B. Leningr. Tekhnol. Inst. im. Lensoveta, Leningrad, USSR. Zhurnal Prikladnoi Khimii (Sankt-Peterburg, Russian Federation) (1971), 44(5), 1068-73.
Abstract
Sorption of Cu ions by silica gel from acetate solns. was studied under 2 different conditions: (a) contact time from 1 min to 24 hr using 4.7 ´ 10-2M Cu(NO3)2 in M AcONa; and (b) contact time from 2 to 120 days with 4.25 ´ 10-4-4.8 ´ 10-2M Cu(NO3)2 in M AcONa. The soly. of the polysilicate salts and the hydrated Cu silicates, in the form of activity products, Kp = aCu2+aSiO32- and Kc = aCu2+aSSiO32-, was (0.8 ± 0.1) ´ 10-19 and 1.9 ´ 10-19, resp.
http://www.reeffrontiers.com/forums/showthread.php?t=15666
http://www.reefland.com/forum/marin...tment/18835-copper-absorbs-into-glass-no.html
and I was asked to comment on a couple of threads at different forums, so I thought I'd also post that info here:
Here are my thoughts on copper:
1. Copper in a reef aquarium is going to be strongly bound to organic materials. That is known to be true in natural seawater at normal copper levels. Maybe when you initially blast seawater with unbound copper there is substantial free copper for some period of time, until the copper concentration eventually drops, or the free copper has a chance to find organics to bind to.
2. While free copper may or may not bind much to glass (and likely none to silicone, IMO), organic bound copper will bind to both glass and silicone surfaces. Not into it, but onto it. It would be an interesting experiment to put some clear cured silicone into a copper/water solution to see if it turns blue. My expectation is that it will not, but I've not done the experiment.
3. It is my opinion that it is not too difficult to remove copper from all plastic and glass objects in an aquarium, but specifically not those that are calcium carbonate. A little bleach will remove organics and copper along with them. A little acid (like vinegar) will remove inorganic copper and some of its precipitates (like copper carbonate or oxide/hydroxide). I think all such objects can be reused after such cleaning.
4. Calcium carbonate surfaces are a much bigger concern. Just as magnesium gets onto and actually into calcium carbonate surfaces, copper and other similar ions would be expected to do the same thing. It is not trivial to clean live rock and sand. In most cases, I'd recommend tossing it. If that is just not an option, cleaning it in acid so that you actually dissolve some of the surface away is a reasonable thing to try, and some folks in my forum have done that.
"Why all the countless years have we not seen dying inverts in tanks where we used copper medications?"
I discuss copper toxicity here:
Tap Water in Reef Aquaria
http://www.advancedaquarist.com/issues/jan2004/chem.htm
from it:
"Toxic Metals in Tap Water: Copper
One of the biggest concerns for aquarists considering the use of tap water is copper. Copper is more toxic to marine invertebrates than it is to humans, so the EPA allows more in tap water than an aquarist would want in an aquarium. Specifically, the EPA has an action level at 1.3 ppm copper in tap water, and it is only a big problem (to the EPA) if more than 10% of the homes on the water supply exceed that level. For that reason, water supplies typically report the 90% level, meaning that 10% of homes exceed that stated value. Many also report the maximum level in any home tested.
Table 5 shows copper levels for a small selection of homes for all of the cities in this study. The reason that copper must be tested at the home tap is that most of the copper in public water supplies comes from the pipes in the home itself. Further, newer does not necessarily mean better, as fresh copper pipes might be even more inclined to release copper to the water than those that have had decades to build up protective coatings.
How much copper is too much? That is difficult to say, and certainly varies from organism to organism. In a recent test,5 Ron Shimek added copper to natural seawater and looked at the effect on sea urchin larvae. He found that concentrations above 10 ppb decreased the larval survival after 48 h, and that concentrations above 100 ppb killed all of them. Whether that translates into particular copper levels to be concerned about in typical reef aquaria is open to discussion. Nevertheless, it points out that copper is potentially toxic at levels well below the EPA action level of 1.3 ppm. That hypothesis is well supported by the literature on copper toxicity.6 Canadian Water Quality Guidelines for the Protection of Aquatic Life suggest that copper be kept to less than a few ppb in fresh water, but they do not provide a salt water recommendation. "
In terms of chemistry of inorganic copper absorption, bare glass is likely to carry a net negative charge in seawater (from silicate) and can bind copper as a copper silicate complex. This is well established in the scientific literature:
Adsorption of some divalent cations from aqueous solution on precipitated silica. Bye, Gerald C.; McEvoy, Michael; Malati, Mounir A. Mid-Kent Coll. Higher Further Educ., Chatham, UK. Journal of the Chemical Society, Faraday Transactions 1: Physical Chemistry in Condensed Phases (1983), 79(10), 2311-18.
Abstract
The adsorption of Co2+, Ni2+, Cu2+, Zn2+, and Cd2+ on the K+ form of a pptd. silica is reported. The adsorption isotherms of Co2+ and of Ni2+ at pH 7.0 and different temps. follow the Langmuir isotherm. The endothermic apparent heats of adsorption, 2 and 10 kJ/mol, resp., are compared with those of Cu2+ and Cd2+. The limiting amt. adsorbed, Xm, of the 4 ions decrease as the pH is lowered. An ion-exchange mechanism between hydrated K+ ions in the outer Helmholtz layer and metal, M2+, ions in soln. is proposed. The results also suggest that (MOH)+ ions are adsorbed at the higher pH values. A plot of Xm at pH 6.0 and 323 K for the 5 ions studied against at. no. exhibits the Irving-Williams sequence. At pH 3.0, i.e. close to the point of zero charge of SiO2, the adsorption results were used to calc. the free energy of specific adsorption, DG, using the Grahame equation. The DG values were -11.1, -10.7, -13.4, -13.3, and -12.1 kJ/mol for Co, Ni, Cu, Zn, and Cd, resp. Specific adsorption is tentatively ascribed to the formation of surface coordination complexes.
Ion-exchange adsorption of zinc and copper ions on silica. Kozawa, A. Univ. Nagoya, Japan. Journal of Inorganic and Nuclear Chemistry (1961), 21(3/4), 315-24.
Abstract
Ion-exchange adsorption of Zn and Cu ions on silica were examd. in 2M NH4Cl contg. ZnO or ZnCl2 and in M NH4OAc contg. 0.05M CuCl2, resp. The ion-exchange adsorption attains equil. in about 2 hrs. at 25°. The adsorption of Zn++ increases with the pH of the soln. up to pH 7.12, then decreases. Between pH 5.48 and 6.38 the mole-ratio of H+ released Zn2+ adsorbed is const. at .apprx.3.0. Zn++ adsorption on heated silica decreases markedly with increase in the heating temp. For Cu++ adsorption, the mole-ratio, H+ rel./Cu2+ ads., is 4.0. The infrared absorption spectra of silica samples, heated silica, and silica treated with Zn2+, Cu2+, and Na+ solns., were taken. A decrease in the absorption intensities for the bending vibration of OH groups on the silica is discussed. A surface complex mechanism for metal ion adsorption of silica is proposed; the silica surface is acting as tridentate ligands for the Zn++ adsorption. A new method for detg. the surface acid and the surface area of silica is suggested.
Reaction of silica gel with copper ions in acetate solutions. II. Rak, R. V.; Dushina, A. P.; Aleskovskii, V. B. Leningr. Tekhnol. Inst. im. Lensoveta, Leningrad, USSR. Zhurnal Prikladnoi Khimii (Sankt-Peterburg, Russian Federation) (1971), 44(5), 1068-73.
Abstract
Sorption of Cu ions by silica gel from acetate solns. was studied under 2 different conditions: (a) contact time from 1 min to 24 hr using 4.7 ´ 10-2M Cu(NO3)2 in M AcONa; and (b) contact time from 2 to 120 days with 4.25 ´ 10-4-4.8 ´ 10-2M Cu(NO3)2 in M AcONa. The soly. of the polysilicate salts and the hydrated Cu silicates, in the form of activity products, Kp = aCu2+aSiO32- and Kc = aCu2+aSSiO32-, was (0.8 ± 0.1) ´ 10-19 and 1.9 ´ 10-19, resp.