Let me first say, this is a project journal. My goal is the sharing of concepts and results. Second, I admit to being a home-built tech junky. This build will be complex and time consuming. It's not my intent to guide anyone else down this path.
As with most projects, this is built on the foundations laid by others. I have always been intrigued by the concept of natural biological filtration systems and I have been attracted to vodka dosing concepts since reading about it some years ago. More recently, I got into a couple of threads on Reef Central: Donovan's Nitrate Destroyer and Nutrient Pulse Reactor DIY DyMiCo system. Just as a recap, these previously mentioned threads utilize organic carbon / vodka dosing into a specially designed reactor.
Most of the working theories available on organic carbon dosing go something like this:
Vodka, sugar, vinegar or some form of organic carbon is dosed into the system to cause bacteria populations to grow exponentially. As bacteria increase exponentially in deep sand beds or inside the surface of live rock, they deplete oxygen and start to consume nitrate to complete their biological processes. The threads above differ from standard vodka dosing by providing an alternate and specific area for this low oxygen condition to occur.
I have been running several variations of the Nutrient Pulse Reactor on a couple of tanks. The difficulty, long term, is keeping the bacteria mat (slime) in check. When I got too aggressive with the organic carbon dosing, the system would eventually clog up and stop functioning properly. Reading Donovan's post, I saw some of the same issues as others tried to implement his system.
Outside of the difficult balancing act, there can be no doubt that these systems work. They will outperform any algae turf scrubber or refugium when it comes to nitrate and phosphate reduction. However, in order to achieve the desirable low nutrient targets, requires either more reactor volume than I have available or more organic carbon than could be dosed, long term. The best balance I could achieve was a system that cycled stably but never reached low nutrient status. Nitrate would hover around 4.0 ppm and phosphate at around .016. When I tried to drive them lower by increasing the carbon dose, the system would clog within weeks.
Consistent / uniform dosing proved to be an issue as well. Every low volume peristaltic pump I tried, even homemade units, didn't perform consistently at anything less than 1 full revolution of the pump. There were always dead spots. This meant that I was diluting the organic carbon so that I could get at least a full revolution on the pump. The catch there is that when you dilute vodka too much, eventually the bacteria creeps into the dilution and consumes the carbon source. This changes the potency, again affecting consistent operation. I even tried vinegar since it is much less potent than vodka but still had to dilute the mix and eventually ended up with bacteria slime and reduced potency in the carbon source.
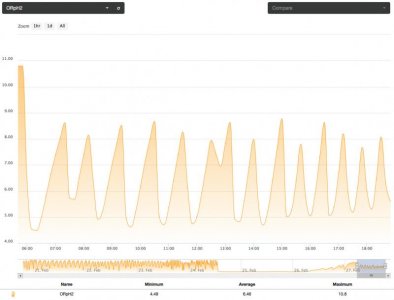
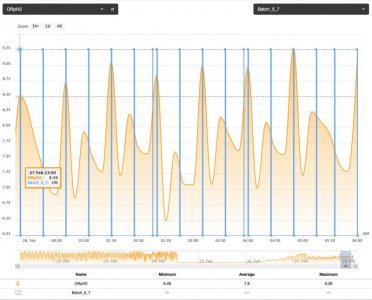
On one such occurrence, I noticed the system was floundering (not cycling normally). I found slime in the bottom of my carbon source. I cleaned the bottle and mixed up a new batch. Then, I turned on the pump to prime the system and got side tracked. When I finally noticed, I had dumped almost a week's worth of carbon into the system. It took about half a day to clear the reactor of the massive dose. The tank suffered no apparent ill effects, other than a little cloudiness but the nitrate went from a consistent 4.0 ppm to 0.25. Phosphate went all the way down to .01 to .02 ppm, from 0.16. All this happened in less than 12 hours. I turned off the carbon dosing and let the system self flush for a few days before starting things back up. My point is to show the potential for affecting these nutrient levels with this method but the following reading, two days later, was also interesting. With the system running a flush cycle every few minutes and no additional carbon going in; the next reading showed. nitrate at 0.5 ppm but phosphate had rebounded to 0.1. This leads me to believe two things.
1. Nitrate is being exported from the system, probably as the theory suggests, as nitrogen gas.
2. Phosphate is not being exported but is being bound up within the increased bacterial population.
If phosphate were actually exported it would build back more slowly when the system was inactive. Since the nitrate was exported from the water, it was building back slowly, as food was added and cycled within the system but phosphate was quickly released back into the water as the bacteria population changed suddenly when I stopped dosing carbon. In fact, I ran a crude test on some of the bacterial slime, adding about 1 to 2 ml to RO water and using the same Red Sea tests and found high levels of both nitrate and phosphate, ten to twenty times higher than the tank water. I also believe that this also may be tied to the theory that live rock can absorb or leach phosphate. Possibly this phenomenon is really related to the bacterial populations within the rock changing. One more point to add is that most vodka dosing regiments require a good skimmer for final nutrient export. The exception is the DiMiCo system, although they do recommend GFO.
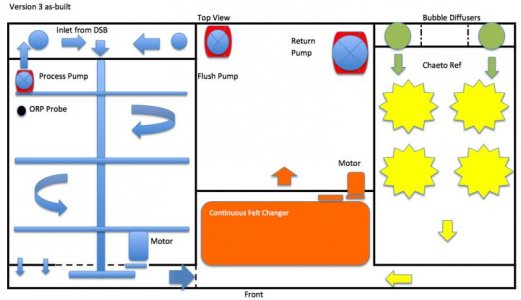
So, I decided to try the third upgrade of my homemade sump to test the feasibility of periodically removing the bio-mat and actually export phosphate while driving the system to it's full potential. I'm calling it the Automatic Biomass Scrubber or ABS for short.
First a little historical perspective...
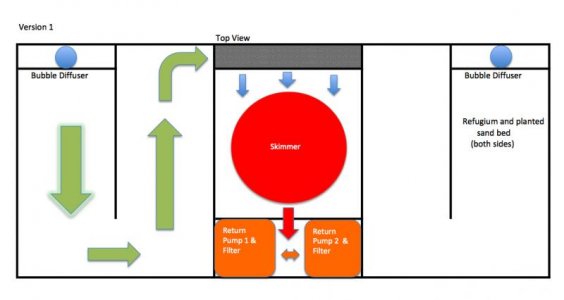
My first sump was a homemade dual refugium with a central protein skimmer. It was 43 X 23 X 19 (external dimensions) and was my first real attempt at sump construction. Each side had a 2" sand bed, planted with caulerpa prolifera. Both sides wound around a baffle to the skimmer chamber and then to the dual return pumps.
The main mistake I made with this system was to place filter sponges in such a way that there was no way for water to flow around or over them when they became clogged. This drove water levels up in the proceeding chambers and required constant attention.
About that time the Roller Mat came out on the US market. I was really ready to get rid of the need to constantly attend to sponges and filter pads and this seemed like the way to go. Of course, I didn't want their prefabricated version, because I thought, yeah, I can do that and I can incorporate it into the sump the way I want it.
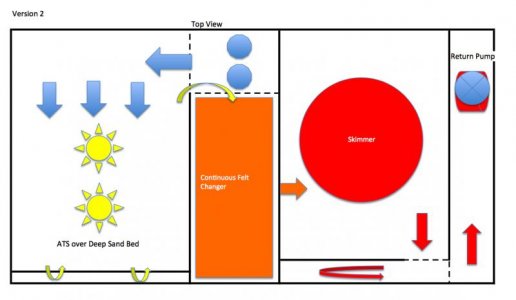
Again, I had the outer "œbox" of the sump built by a local plastics company due to the size and concern over leaks. I always do this. I am just not set up, with the tools and jigs to handle the long seams without leak concerns. I had the new box built with ¼ in plexiglass, again keeping with relatively the same footprint (43 X 23). I went an inch shorter on the depth this time, to only 18 inches deep. I decided, with all the internal baffles and bracing, I did not need to have anything thicker than ¼. This sump has been up and running for a couple of years now without issue. The primary method of nutrient export was a sand bed in four stacked trays under an algae turf scrubber. I also have a Reef Octopus protein skimmer. The continuous felt changer was between the ATS and skimmer.
This system ran for about a year prior to my interest in the system described in the Nutrient Pulse Reactor post.
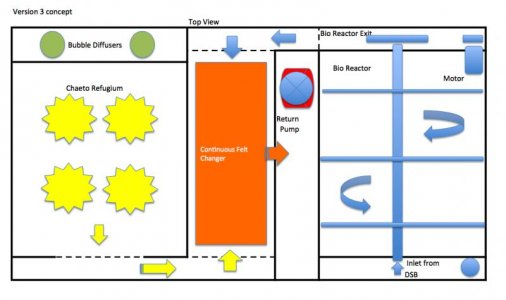
This new ABS system will be composed of 4 primary components:
Remote Deep Sand Bed
Bio Reactor with motorized agitation
Rotating Chaeto Reactor / Refugium
Continuous Felt Changer
The first thing to keep in mind is: this ABS side is not operated as a continuous-flow system. This means water does not flow through this section continuously. It is a batching system. Water is pumped into the system, carbon is dosed and then a wait period allows for the reaction to occur. ORP is used to determine when the process is complete and then another batch is processed. This was the primary reason for combining the chaeto refugium; to have a parallel section with continuous flow. The chaeto will also hopefully increase the pH as well.
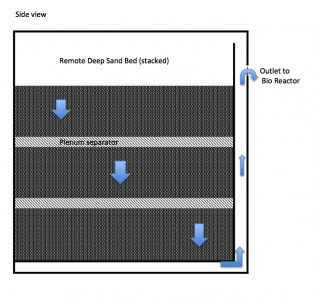
I'm trying to reuse as many pre-existing components as possible. For example, I removed the baffles and structures from my original (ver 1) 43x23x19 sump and will build the new components into this box. Also, the Remote Deep Sand Bed will be constructed from a 24x24x6 box, from a previous project. This box is vertical so it is 24 inches deep and long but only 6 inches wide. It will fit above the sump, between the back of the tank and the wall.
Locating the DSB away from the Bio Reactor solves two problems. First, it provides for fluid separation between the sand bed and Bio Reactor and it's carbon source. I had issues with other versions of this system due to the close proximity of the two. Organic carbon would migrate into the sand bed and the resulting bio mat would quickly clog it. Secondly, locating the DSB away from the Bio Reactor gave me more reactor space and allows me to reuse these existing boxes. The sand bed will also need to be layered with a plenum space between the three sand layers. I added a bottom panel, resting on two 3/8 inch supports with ¼ inch holes and a baffle on one end. A shower-head cover will diffuse the force of each cycle, preventing the sand from being disturbed. I've used this concept before to diffuse the water into a compartment. It works well.
Each cycle or batch of water will then be directed down through each layer, exiting through the bottom panel and out the side, on it's way to the bio-reactor. The concept here is to provide a place for nitrification to take place prior to being pushed to the bio-reactor for de-nitrification. Then on a weekly basis, I will turn on the rotating arms in the bio reactor and flush out any loosely attached bacterial slime into the CFC for removal. The CFC will also capture and remove and loose pieces of cheato.
As with most projects, this is built on the foundations laid by others. I have always been intrigued by the concept of natural biological filtration systems and I have been attracted to vodka dosing concepts since reading about it some years ago. More recently, I got into a couple of threads on Reef Central: Donovan's Nitrate Destroyer and Nutrient Pulse Reactor DIY DyMiCo system. Just as a recap, these previously mentioned threads utilize organic carbon / vodka dosing into a specially designed reactor.
Most of the working theories available on organic carbon dosing go something like this:
Vodka, sugar, vinegar or some form of organic carbon is dosed into the system to cause bacteria populations to grow exponentially. As bacteria increase exponentially in deep sand beds or inside the surface of live rock, they deplete oxygen and start to consume nitrate to complete their biological processes. The threads above differ from standard vodka dosing by providing an alternate and specific area for this low oxygen condition to occur.
I have been running several variations of the Nutrient Pulse Reactor on a couple of tanks. The difficulty, long term, is keeping the bacteria mat (slime) in check. When I got too aggressive with the organic carbon dosing, the system would eventually clog up and stop functioning properly. Reading Donovan's post, I saw some of the same issues as others tried to implement his system.
Outside of the difficult balancing act, there can be no doubt that these systems work. They will outperform any algae turf scrubber or refugium when it comes to nitrate and phosphate reduction. However, in order to achieve the desirable low nutrient targets, requires either more reactor volume than I have available or more organic carbon than could be dosed, long term. The best balance I could achieve was a system that cycled stably but never reached low nutrient status. Nitrate would hover around 4.0 ppm and phosphate at around .016. When I tried to drive them lower by increasing the carbon dose, the system would clog within weeks.
Consistent / uniform dosing proved to be an issue as well. Every low volume peristaltic pump I tried, even homemade units, didn't perform consistently at anything less than 1 full revolution of the pump. There were always dead spots. This meant that I was diluting the organic carbon so that I could get at least a full revolution on the pump. The catch there is that when you dilute vodka too much, eventually the bacteria creeps into the dilution and consumes the carbon source. This changes the potency, again affecting consistent operation. I even tried vinegar since it is much less potent than vodka but still had to dilute the mix and eventually ended up with bacteria slime and reduced potency in the carbon source.
On one such occurrence, I noticed the system was floundering (not cycling normally). I found slime in the bottom of my carbon source. I cleaned the bottle and mixed up a new batch. Then, I turned on the pump to prime the system and got side tracked. When I finally noticed, I had dumped almost a week's worth of carbon into the system. It took about half a day to clear the reactor of the massive dose. The tank suffered no apparent ill effects, other than a little cloudiness but the nitrate went from a consistent 4.0 ppm to 0.25. Phosphate went all the way down to .01 to .02 ppm, from 0.16. All this happened in less than 12 hours. I turned off the carbon dosing and let the system self flush for a few days before starting things back up. My point is to show the potential for affecting these nutrient levels with this method but the following reading, two days later, was also interesting. With the system running a flush cycle every few minutes and no additional carbon going in; the next reading showed. nitrate at 0.5 ppm but phosphate had rebounded to 0.1. This leads me to believe two things.
1. Nitrate is being exported from the system, probably as the theory suggests, as nitrogen gas.
2. Phosphate is not being exported but is being bound up within the increased bacterial population.
If phosphate were actually exported it would build back more slowly when the system was inactive. Since the nitrate was exported from the water, it was building back slowly, as food was added and cycled within the system but phosphate was quickly released back into the water as the bacteria population changed suddenly when I stopped dosing carbon. In fact, I ran a crude test on some of the bacterial slime, adding about 1 to 2 ml to RO water and using the same Red Sea tests and found high levels of both nitrate and phosphate, ten to twenty times higher than the tank water. I also believe that this also may be tied to the theory that live rock can absorb or leach phosphate. Possibly this phenomenon is really related to the bacterial populations within the rock changing. One more point to add is that most vodka dosing regiments require a good skimmer for final nutrient export. The exception is the DiMiCo system, although they do recommend GFO.
So, I decided to try the third upgrade of my homemade sump to test the feasibility of periodically removing the bio-mat and actually export phosphate while driving the system to it's full potential. I'm calling it the Automatic Biomass Scrubber or ABS for short.
First a little historical perspective...
My first sump was a homemade dual refugium with a central protein skimmer. It was 43 X 23 X 19 (external dimensions) and was my first real attempt at sump construction. Each side had a 2" sand bed, planted with caulerpa prolifera. Both sides wound around a baffle to the skimmer chamber and then to the dual return pumps.
The main mistake I made with this system was to place filter sponges in such a way that there was no way for water to flow around or over them when they became clogged. This drove water levels up in the proceeding chambers and required constant attention.
About that time the Roller Mat came out on the US market. I was really ready to get rid of the need to constantly attend to sponges and filter pads and this seemed like the way to go. Of course, I didn't want their prefabricated version, because I thought, yeah, I can do that and I can incorporate it into the sump the way I want it.
Again, I had the outer "œbox" of the sump built by a local plastics company due to the size and concern over leaks. I always do this. I am just not set up, with the tools and jigs to handle the long seams without leak concerns. I had the new box built with ¼ in plexiglass, again keeping with relatively the same footprint (43 X 23). I went an inch shorter on the depth this time, to only 18 inches deep. I decided, with all the internal baffles and bracing, I did not need to have anything thicker than ¼. This sump has been up and running for a couple of years now without issue. The primary method of nutrient export was a sand bed in four stacked trays under an algae turf scrubber. I also have a Reef Octopus protein skimmer. The continuous felt changer was between the ATS and skimmer.
This system ran for about a year prior to my interest in the system described in the Nutrient Pulse Reactor post.
This new ABS system will be composed of 4 primary components:
Remote Deep Sand Bed
Bio Reactor with motorized agitation
Rotating Chaeto Reactor / Refugium
Continuous Felt Changer
The first thing to keep in mind is: this ABS side is not operated as a continuous-flow system. This means water does not flow through this section continuously. It is a batching system. Water is pumped into the system, carbon is dosed and then a wait period allows for the reaction to occur. ORP is used to determine when the process is complete and then another batch is processed. This was the primary reason for combining the chaeto refugium; to have a parallel section with continuous flow. The chaeto will also hopefully increase the pH as well.
I'm trying to reuse as many pre-existing components as possible. For example, I removed the baffles and structures from my original (ver 1) 43x23x19 sump and will build the new components into this box. Also, the Remote Deep Sand Bed will be constructed from a 24x24x6 box, from a previous project. This box is vertical so it is 24 inches deep and long but only 6 inches wide. It will fit above the sump, between the back of the tank and the wall.
Locating the DSB away from the Bio Reactor solves two problems. First, it provides for fluid separation between the sand bed and Bio Reactor and it's carbon source. I had issues with other versions of this system due to the close proximity of the two. Organic carbon would migrate into the sand bed and the resulting bio mat would quickly clog it. Secondly, locating the DSB away from the Bio Reactor gave me more reactor space and allows me to reuse these existing boxes. The sand bed will also need to be layered with a plenum space between the three sand layers. I added a bottom panel, resting on two 3/8 inch supports with ¼ inch holes and a baffle on one end. A shower-head cover will diffuse the force of each cycle, preventing the sand from being disturbed. I've used this concept before to diffuse the water into a compartment. It works well.
Each cycle or batch of water will then be directed down through each layer, exiting through the bottom panel and out the side, on it's way to the bio-reactor. The concept here is to provide a place for nitrification to take place prior to being pushed to the bio-reactor for de-nitrification. Then on a weekly basis, I will turn on the rotating arms in the bio reactor and flush out any loosely attached bacterial slime into the CFC for removal. The CFC will also capture and remove and loose pieces of cheato.