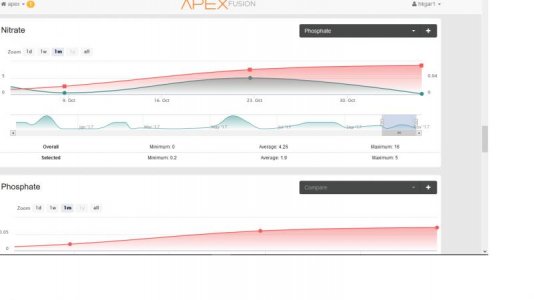
Yes. Its redfield ratio of NWhile the real chemists can chime in and correct me, I wonder if the tank is phosphate limited and not allowing nitrates to come down naturally. My recent research shows shooting for a ratio of 10ppm N to .10ppm P seems to keep everything happy and in check. Assuming the Phosphate reading is correct (.04 falls within the acceptable error range for some readers) the tank could actually be at 0 phosphates. I am learning the hard way that a consistent reading of 0 phosphates isn't a good thing. At least not for me and my system (and a lot of others too.)
Sent from my SM-G930F using Tapatalk