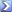What I see here is aquarists coming at this topic from different angles with different objectives. Looking at the extremes:
Person 'A' wants to emulate an ocean reef's oligotrophic water. To keep Acropora and similar corals in such an environment, they need to be fed a relatively large amount and variety of high quality foods. Obviously, to maintain these pristine water conditions, nutrient export needs to be very efficient and removal of uneaten food/detritus as soon as possible, before it breaks down, is a top priority. This person is not concerned with 'all the little critters' in this case, since the focus is on the corals.
Person 'B' wants to have as complete an ecosystem as possible. He finds delight in seeing all the worms, crustaceans, etc. moving around the tank looking for tidbits to eat. He wouldn't dream of vacuuming up these creatures and throwing them out. Fish, coral and other creatures complete this little ecosystem and he's happy to let all the small benthic animals chow through the detritus until it's been turned to near inert mulm.
IMO, there is no 'right' or 'wrong' here, just 'different' as long as the tank maintains the desired level of eutrophication that the reef keeper finds acceptable and the system can effectively deal with. Problems result when the degree of eutrophication becomes damaging to the life forms we are trying to keep and the system as a whole becomes unbalanced.
i thought i have been making this clear throughout this thread. i have mentioned many times that the amount of nutrient export needs to match the trophic level of the system wanting to be emulated. a substrate makes perfect sense in those systems maintaining those organism from more eutrophic areas. a system without a substrate and emphasis detrital removal makes more sense for those emulating the more oligotrophic area. there of course is some leeway, but the more you stray form the more resources that will be necessary to maintain the desired trophic state. resins, carbon dosing, GFO are all masking the affects of eutrophication by going after the iP in the water column.
confused. this graph is of the marine environment, ....done by bacterial activity. the same activity that allows "cooking" of LR. the phosphate solubilizing bacteria are doing this.
The graph is of a mangrove swamp. Acidification and other process are just as likely to cause dissolution at more significant levels there than a reef tank than some unidentified solubilizing bacteria may or /may not.
and what causes this acidification? what causes the pH to drop? i give you hint. look up decomposition of organic material and what is one of its by products.
"cooking", purging, or curing LR utilize this process.
Mineralization or breakdown to refractory organics /aka sinking phosphate seem more likely, imo.
"Cooking" usually means leaving the rock in a darkened bin of water for a year or so .I think it may work as a result of bacterial degradation and mineralization ,sinking the phosphate and other elements in refractory organics or via mineralization. If it is a process that simply liberates the phosphate as you suggest via unidentified solubilizing bacteria with no export then where do you suppose the phosphate goes in the end?
it becomes the detritus you see at the bottom of the tank in which you are "cooking" the LR in. what else do you think that detritus is comprised of?
Waste.
Look at it this way. You toss a hamburger into the tank, your fish eat it and poop out 80% as undigested organic matter. The sand bed critters eat that remaining .8 of a burger and poop out .64 of a hamburger. That .64 of a hamburger gets consumed again and .51 of a burger is pooped out. .51 of a burger is consumed again and .4 is pooped out. Only 10 times through and you only have a bite (10%) left. It's an orgy of pooped out hamburger gorging until there is nothing left.

That is why you want a healthy benthic community in your tank.
That is why you do not want to stir your sand bed, in effect grinding up all those beneficial organisms and turning them into a nutrient source instead of a source of nutrient consumption.
no offense, but you really do not understand the basics of a biological system. please look at how a sewage treatment plant operates because i have a feeling you feel that they also perform magic and are also able to make P vanish.
in your example all of your organisms are operating at higher than 100% efficiency. i would wish this were true. if what you say were true, then that ONE organism must increase its mass by the amount of material taken up by the material taken in that is not expelled. that would mean that that ONE organism would be quite large and getting bigger. for every ONE organism you add you must also add in the amount of food and waste created.
lets look at your example more closely.

Look at it this way. You toss a hamburger into the tank, your fish eat it and poop out 80% as undigested organic matter.
so that ONE organism is increasing at a rate of 20% of its incoming nutrients. for every meal that ONE organism is increasing in it mass. are you increasing in your mass at a rate of 20% of the mass of your incoming food?
The sand bed critters
here is your problem. you can not lump them together that way. you need to look at the individual organism. for every mouth you add, you add that organisms waste. for that one additional organism that is alive, you must have food for that organisms, or it would not be alive. eat that remaining .8 of a burger and poop out .64 of a hamburger. That .64 of a hamburger gets consumed again and .51 of a burger is pooped out. .51 of a burger is consumed again and .4 is pooped out. Only 10 times through and you only have a bite (10%) left. It's an orgy of pooped out hamburger gorging until there is nothing left.
 there can not be nothing left. that is the problem and i think that is where you are missing what P can and can not do. P has to be exported in order for it to leave the system. whether or not it is in an organism, in the new food coming into the system, or in the waste organic material in, it is still IN THE SYSTEM. all of those extra organisms are telling you this.
there can not be nothing left. that is the problem and i think that is where you are missing what P can and can not do. P has to be exported in order for it to leave the system. whether or not it is in an organism, in the new food coming into the system, or in the waste organic material in, it is still IN THE SYSTEM. all of those extra organisms are telling you this.
That is why you want a healthy benthic community in your tank.
and that is why biodiversity and ecosystem are fancy words for phosphate sinks (cesspool)
That is why you do not want to stir your sand bed, in effect grinding up all those beneficial organisms and turning them into a nutrient source instead of a source of nutrient consumption.
that is why i suggest siphoning up all of that waste organic material and those pesky benthic organisms and export all of that P
did you understand my hamburger example earlier about the amount of hamburgers go through an organism in a given period? all of the resources needed for an organism to survive must be available in order for the organism to survive.
Personally, I advocate against disturbing a sand bed out of concern for the fauna themselves, given how important they are in my particular approach to reefing. I don't think anyone really knows what effect vacuuming would have on them, but I err on the side of caution. I think there's also something there about stirring up pockets of reduced substances which would then draw down oxygen levels, but that's also speculative until more research is done.
if the system is wanting to be more eutrophic, then absolutely keep the benthic organisms, or if you really like them around. that is the your purpose. the problem i have is that all of these organisms are not doing anything to help maintain the trophic state of an oligotrophic system.
this happen to the benthic organisms if one were to siphon the substrate on a regular basis.

they would be siphoned up and removed along with the waste organic material that is at the bottom of the bucket.

as the waste organics start building up again in the substrate, they will again repopulate. this is no different that what happens when someone starts up a new system.
--------------------------
i want to clarify what i am suggesting about nutrient exports. i am not saying that detritus removal is the only way to remove P. i think that it should be made primary instead of tertiary. the way the marine hobby goes about P removal is more top down. i am just suggesting from the bottom up, that is all. i suggest using detritus removal by water changes as primary, with a heavy dose of skimming. if that is also not adequate then use resins, GFO, carbon dosing if needed. all of this is of course matched to the trophic state wishing to be emulated. adjust the skimming, the feeding, the siphoning, etc..... to maintain that particular trophic state. the way it is done now is the opposite. the only time detritus removal seems to be recommended is when the system has become so eutrophic that the only recourse is to start over. why does starting over help if it is not bringing the system back to as low of an initial P as possible?
i wanted to post some information about decomposition and fertilizers.
Chapter 2. Organic matter decomposition and the soil food web
Chapter 1, The Decomposition Process
The Major Biogeochemical Cycles and Their Interactions
note the production of CO2 by the microorganism.
The Best Animal Manure Fertilizers
quote from above link:
"The best animal manure fertilizers come from livestock, fish, worms and even bats."
which of those organisms are in our system?
 Fish-Waste Composting
Fish-Waste Composting
G~