oh damn i just missed a chance to send a sample as i had guests from maryland  this world is too dam*n small if you ask me
this world is too dam*n small if you ask me 
next time they come they might bring you some as long as i have not won the battle, and it does not look good.
lights out and no waterchanges killed almost all of my sps, with only the green monti digi surviving. all others, including the gorgonians which are affected worst, still make it and i have just finished another 2 days without light. lets see where this is going... still dosing microbe-lift special blend accordingly.
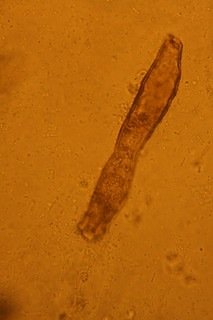
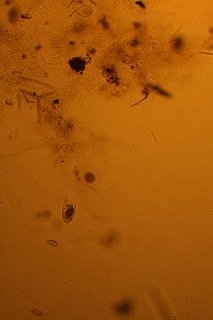
i also had a look at them through my fathers microscope, although i was only able to do 20x, i am pretty sure they are dinos as they almost look like your avatar, pants! will get some new objectslides for the scope and might invest in an eyepiece cam, or is there a cheaper way to take microscopic photos? that way i could share them here...
next time they come they might bring you some as long as i have not won the battle, and it does not look good.
lights out and no waterchanges killed almost all of my sps, with only the green monti digi surviving. all others, including the gorgonians which are affected worst, still make it and i have just finished another 2 days without light. lets see where this is going... still dosing microbe-lift special blend accordingly.
i also had a look at them through my fathers microscope, although i was only able to do 20x, i am pretty sure they are dinos as they almost look like your avatar, pants! will get some new objectslides for the scope and might invest in an eyepiece cam, or is there a cheaper way to take microscopic photos? that way i could share them here...