You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Kalk Reactor for pH control
- Thread starter t.trezona
- Start date
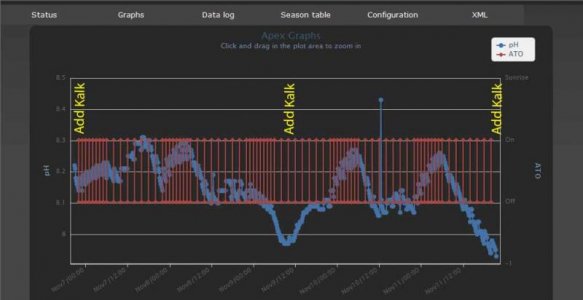
I have been using my kalk reactor for pH control. Adds about as much kalk as evaporative losses and keeps pH perfectly stable. Why don't more people use this method?
I do and mine zig zags all day...
Attachments
I've dosed it from a settled reservoir for about 15 years or so. I prefer that to reactors since it is easier to control the potency and let undesirtable solids settle out. It is the sole source of supplemental calcium and alkalinity in my tank. :
Randy would you please expand on your comments. I use a Reactor I built. The kalk is mixed for 1 minute every hour in a 5 gal vessel. The intake for the mixing pump is about half way up the vessel and the output I have directed toward the base. There is only what appears to be clear saturated solution which is pumped out the top of the vessel. Why would the potency change and why would I be delivering contaminants? What would they be?
Many folks find that their reactors do not deliver fully saturated limewater when they actually look closely. That will, of course, depend on how often you stir and the nature of the design.
When stirring, many reactors (not all) have the potential to deliver some of the powder, which will contain undesirable impurities to some extent. These include things like copper and phosphate, if present in the lime solids. Whether that is important or not may depend on the purity of the lime solids you use.
It is also not technically easy to mix vinegar into the limewater when using a reactor, if this is a route you choose to go to boost the potency of the limewater.
When stirring, many reactors (not all) have the potential to deliver some of the powder, which will contain undesirable impurities to some extent. These include things like copper and phosphate, if present in the lime solids. Whether that is important or not may depend on the purity of the lime solids you use.
It is also not technically easy to mix vinegar into the limewater when using a reactor, if this is a route you choose to go to boost the potency of the limewater.
I've never considered the possibility that my Kalk would not be fully saturated all the time, except of course when the CaOH runs out. Nor would it make any difference using it strictly for pH control. It would simply add a little extra water. I check the resovoir daily for CaOH and recharge when low, which is about every 2 months or so. I'm certain there is some ppt that comes over on occasion when the s mix cycle happens near a dose time. Could this be significant? Would there be any means of checking this? I know my PO4 is minimal. I don't know about the copper. My animals all seem to be very healthy. Coral growth is rapid ect."
disc1
-RT * ln(k)
Nor would it make any difference using it strictly for pH control. It would simply add a little extra water.
And calcium and alkalinity. Dosing strictly by pH numbers sound like a recipe to get alk all out of whack.
If I run the Kalk reactor and my Ca reactor I am stable at about 8 dKH with calcium of 450. In order to boost the DKH I have added Randy's recipe 1. If I increase the CO2 flow on my Ca reactor just slightly (currently I run the pH at 6.80) the volume of water added to maintain my tank pH is more than my evaporative losses.
Nor would it make any difference using it strictly for pH control.
Assuming it met that need, that's correct. many people, hwoever, do not get enough pH control, even with fully saturated limewater replacing all evaporated water, which limts the dosing possible.
I'm certain there is some ppt that comes over on occasion when the s mix cycle happens near a dose time. Could this be significant? Would there be any means of checking this? I know my PO4 is minimal. I don't know about the copper. My animals all seem to be very healthy. Coral growth is rapid ect."
I do not think a little solids is enough concern to warrant doing anything. It is jjust a potenntial drawback relative to dosing from a settled reservoir. Dosing from a reservoir takes a lot of space somewhere, but otherwise is probably cheaper than a reactor as well. So unless space is a concern, I'd opt for a reservoir. If you already have a reactor, by all means keep it.
Assuming it met that need, that's correct. many people, hwoever, do not get enough pH control, even with fully saturated limewater replacing all evaporated water, which limts the dosing possible.
I'm certain there is some ppt that comes over on occasion when the s mix cycle happens near a dose time. Could this be significant? Would there be any means of checking this? I know my PO4 is minimal. I don't know about the copper. My animals all seem to be very healthy. Coral growth is rapid ect."
I do not think a little solids is enough concern to warrant doing anything. It is jjust a potenntial drawback relative to dosing from a settled reservoir. Dosing from a reservoir takes a lot of space somewhere, but otherwise is probably cheaper than a reactor as well. So unless space is a concern, I'd opt for a reservoir. If you already have a reactor, by all means keep it.
Why so worried about pH?
Alk is the important metric. If alk is fine why worry?
This has always concerned me. What is the pH fluctuation on a reef in the south pacific. I'd say none to minimal. I don't know what the ability of a coral is to adjust (maintain) their intracellular pH is when the tank fluctuation goes from say 8.15 during the day to 7.80 at night. Does anybody know this? Seems to me it would be a significant stress. I don't know for sure but it has always concerned me enought to want to maintain my pH constant. Happy to hear your thoughts.
The pH moves around quite a bit in lagoons, but it is a fine goal to keep the pH from dropping below about 8.1 to 8.2.
disc1
-RT * ln(k)
I don't think people understand how small of a change 8.2 to 7.8 is in pH. Intracellular pH is a very important thing and most of the animals we keep run an intracellular pH that is well below what the water around them is. They are well adapted to maintain that gradient, and I don't think a few points swing on pH is going to mess them up. Surely with all the currents blowing around on the reef and temperature and weather the pH fluctuates to some extent.
If we had an intracellular pH change of 0.6 we would be very dead. A blood pH change of 0.6 will kill most people let alone an intracellular change of that much. That doesn't happen with us because we have tremendously powerful systems (multiple different types of buffering systems as well as an ability to export or hang on to acids, bases, and CO2) for maintaining our pH. Organisms that have evolved in an environment where the pH basically never changes do not have sophisticated mechanisms for maintaining pH and therefore a 0.6 fluctuation will definitely stress them. I'm not saying they can't tolerate that change, they can. Its just a significant stress for them. Thats why I prefer to maintain a stable pH in my system. I don't have clinical trial data with corals. I'm guessing. But I suspect I'm correct.
Well, I think it's true that some stony corals would grow more quickly if the pH were consistently 8.4 or so, as opposed to 7.8, but we see a lot of very nice tanks that seem to run more like 7.8. Corals, and all organisms of which I'm aware, have a lot of mechanisms to control their internal pH. I agree that it's possible that lower pH might cause some stress, but I have a hard time believing that it's a critical issue, given that so many tanks have pH levels that vary significantly during the day, yet manage to do so well.